A Review Article on Development of Forced Degradation and Stability Indicating Studies for Drug Substance and Drug Product

Impact from the Recent Issuance of ANVISA Resolution RDC-53/2015 on Pharmaceutical Small Molecule Forced Degradation Study Requirements | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology

Forced degradation studies for Drug Substances and Drug Products- Scientific and Regulatory Considerations | Semantic Scholar

The Degradation Map Process – A Tool for Obtaining a Lean Stability Strategy in Drug Development - Journal of Pharmaceutical Sciences

Degradation conditions for pharmaceutical drugs in forced degradation... | Download Scientific Diagram

FORCED DEGRADATION STUDIES: PRACTICAL APPROACH - OVERVIEW OF REGULATORY GUIDANCE AND LITERATURE FOR THE DRUG PRODUCTS AND DRUG SUBSTANCES | Semantic Scholar

Forced Degradation Studies for Drug Substances and Drug Products - A Regulatory Considerations - Veeprho
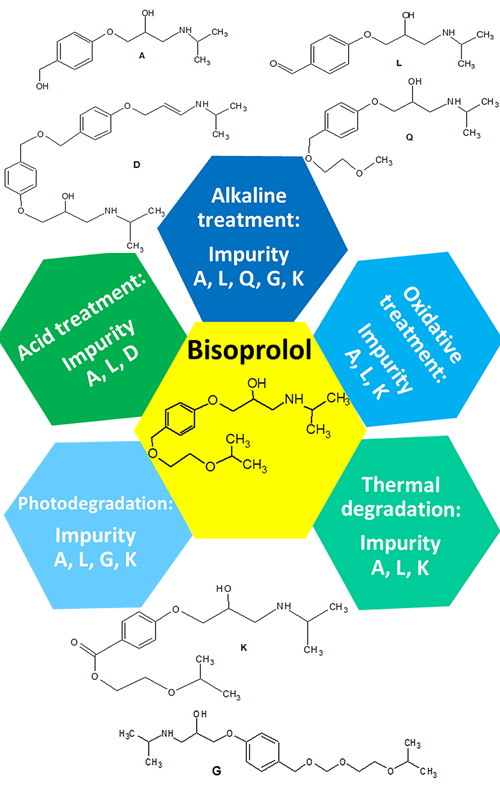
Forced degradation studies and structural characterization of related substances of bisoprolol fumarate in finished drug product using LC–UV–MS/MS | Journal of the Serbian Chemical Society

Overview on Development and Validation of Force degradation studies with Stability Indicating Methods. – Biosciences Biotechnology Research Asia

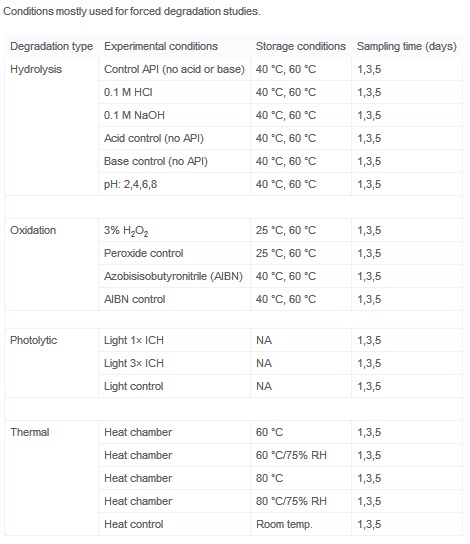
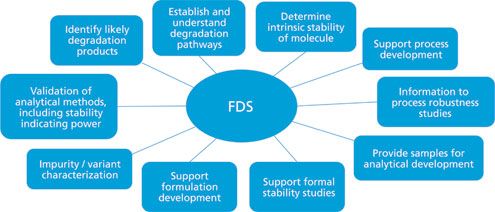
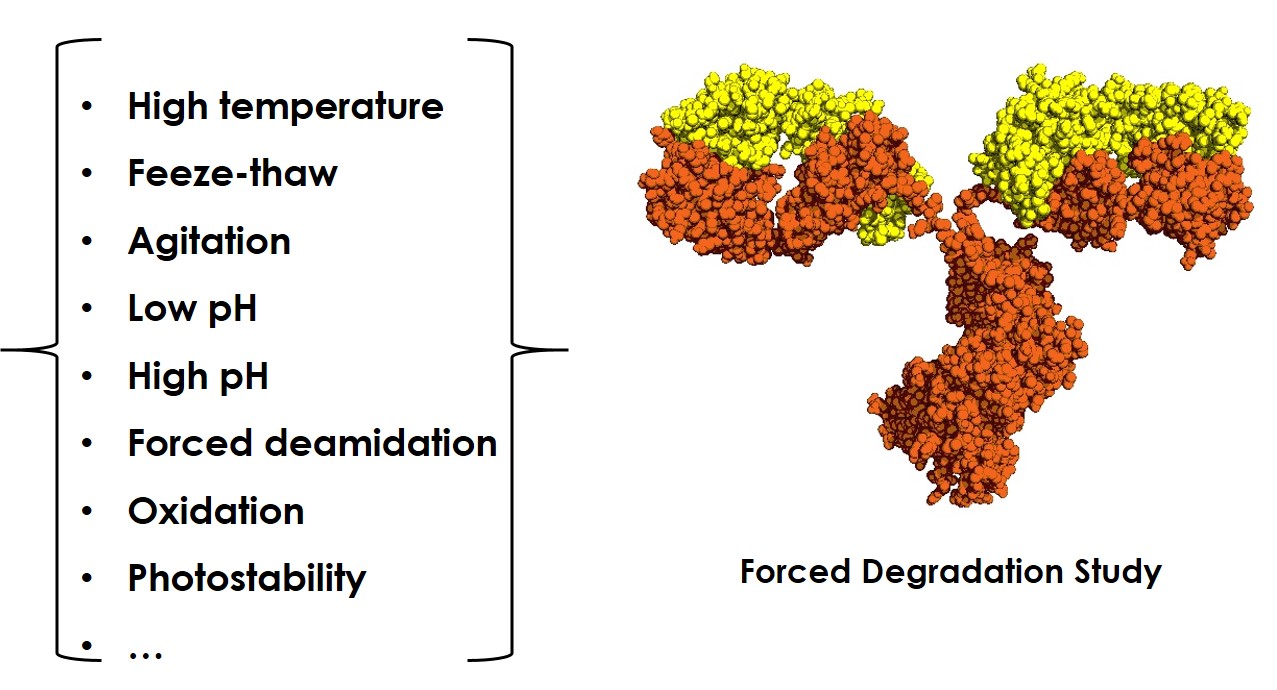
Development of forced degradation and stability indicating studies of drugs—A review - ScienceDirect

Forced Degradation to Develop Stability-indicating Methods | Pharmaceutical Outsourcing - The Journal of Pharmaceutical & Biopharmaceutical Contract Services