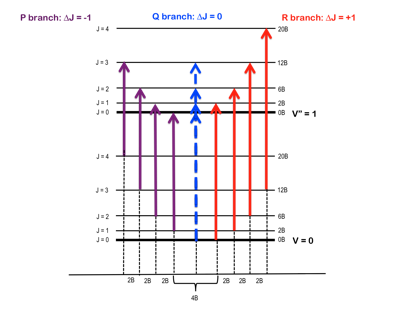
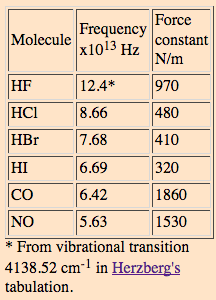
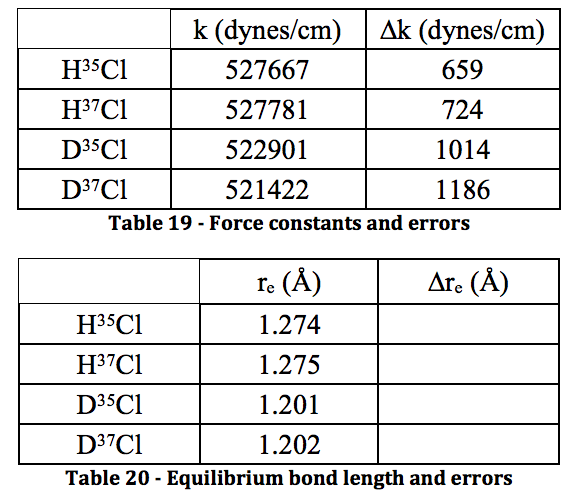
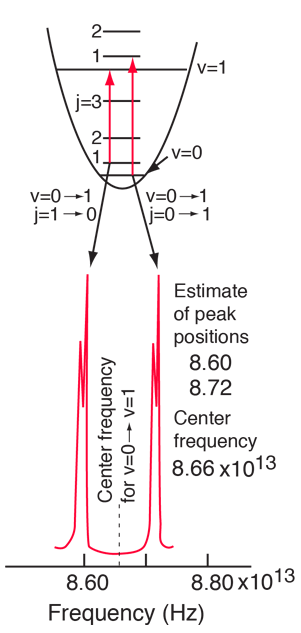
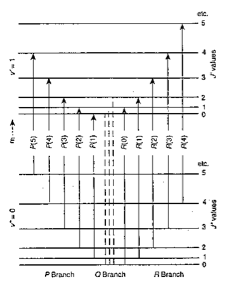
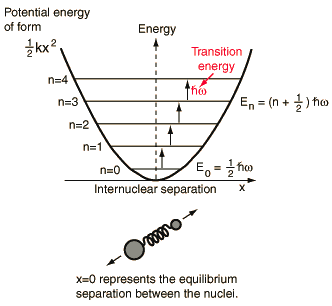
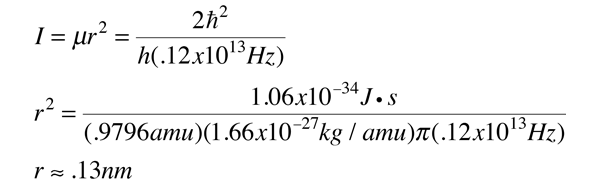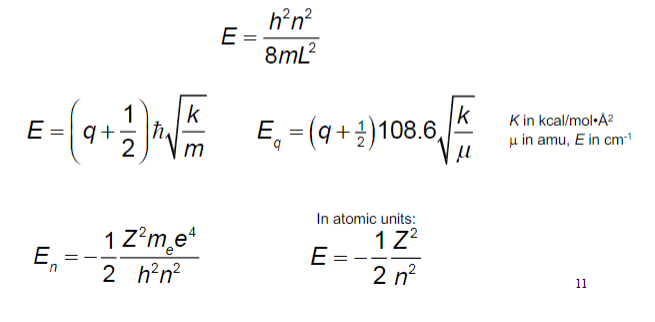The moment of inertia of `HCl` molecule about an axis passing through its centre of mass and - YouTube
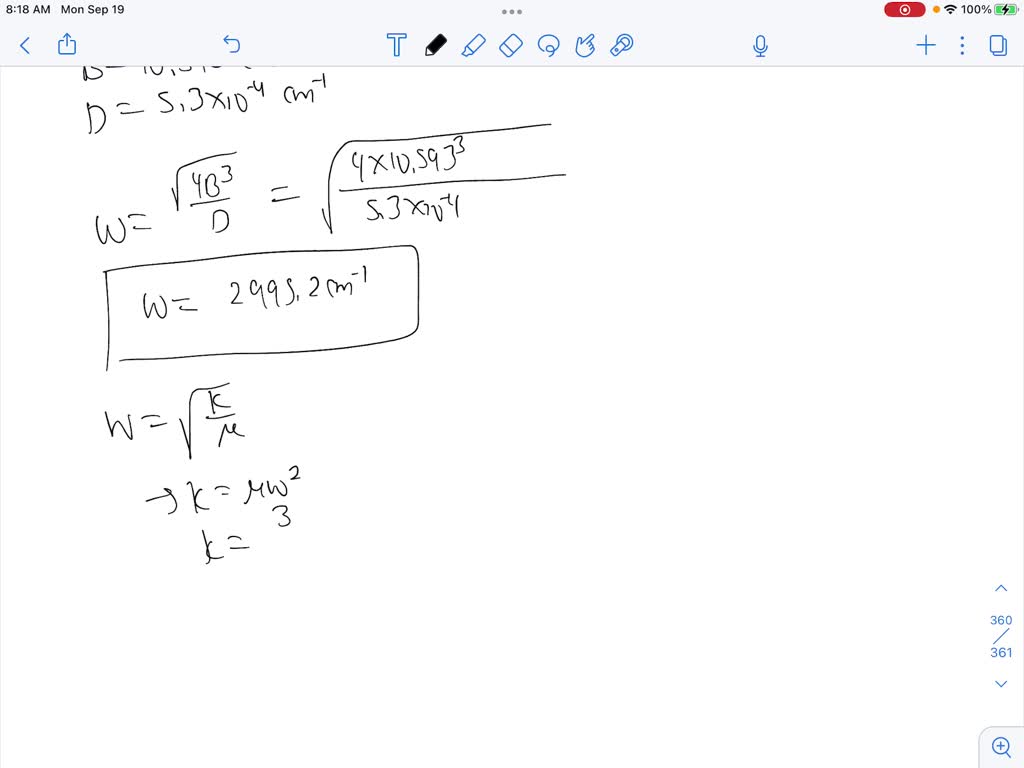
SOLVED: The diatomic molecule HCI has a B value of 10.593 cm and a centrifugal distortion constant Dof 5.3 X 10-4 cm [ Estimate the vibrational frequency and force constant of the
H-X force constant f r (in mdyn/ ˚ A) for the hydrogen halides as a... | Download Scientific Diagram

OneClass: Consider a diatomic molecule such as HCl. The fundamental vibrational frequency has been me...

SOLVED: Calculate the force constant (use proper unit) of HCl (2990) where the number in the parenthesis is the vibrational wavenumber in cm-1.

SOLVED: (1) Given that the spacing between vibrational energy levels of the HCl molecule is 0.36 eV, calculate the effective force constant. (2) Find the classical temperature associated with this difference between