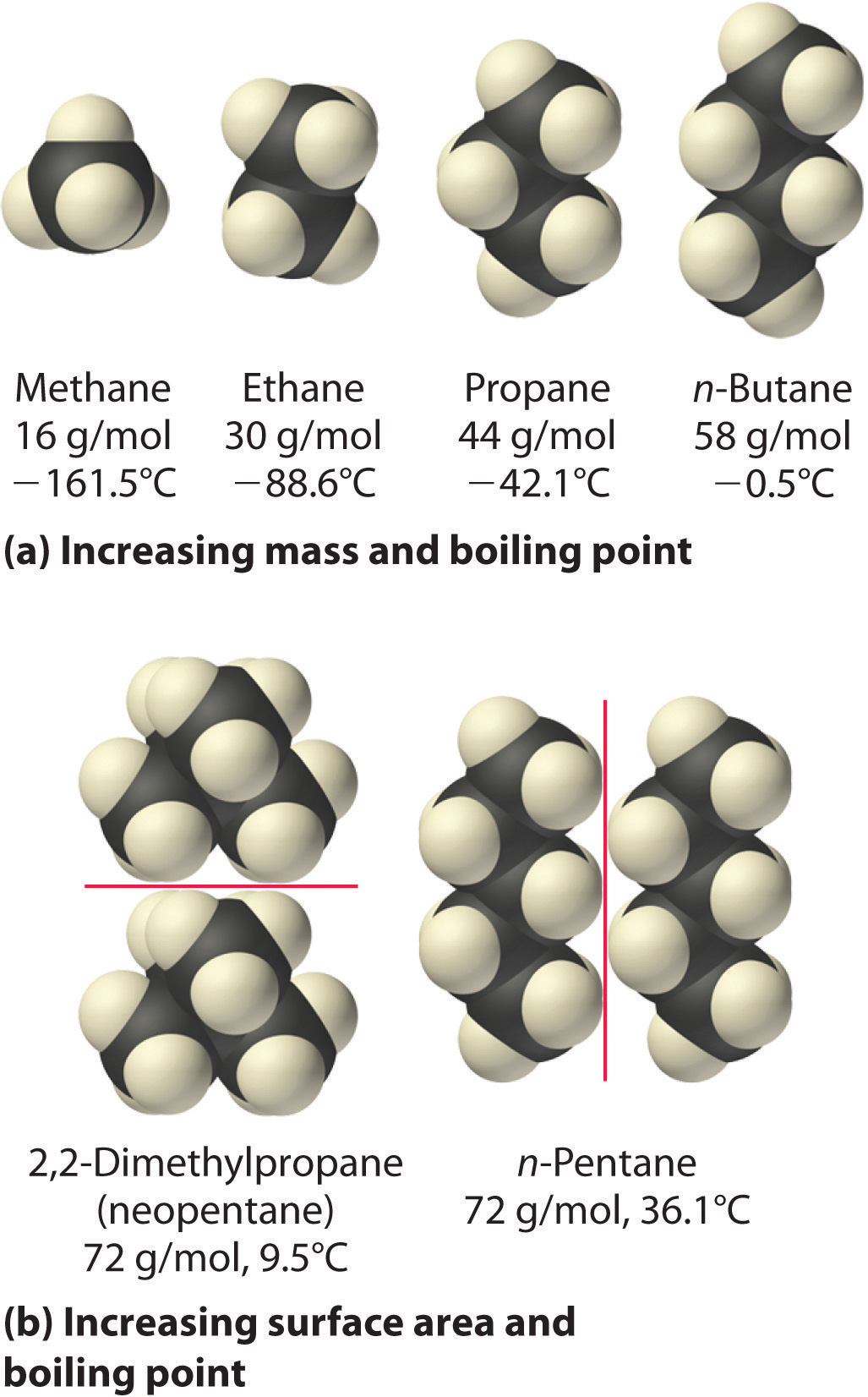
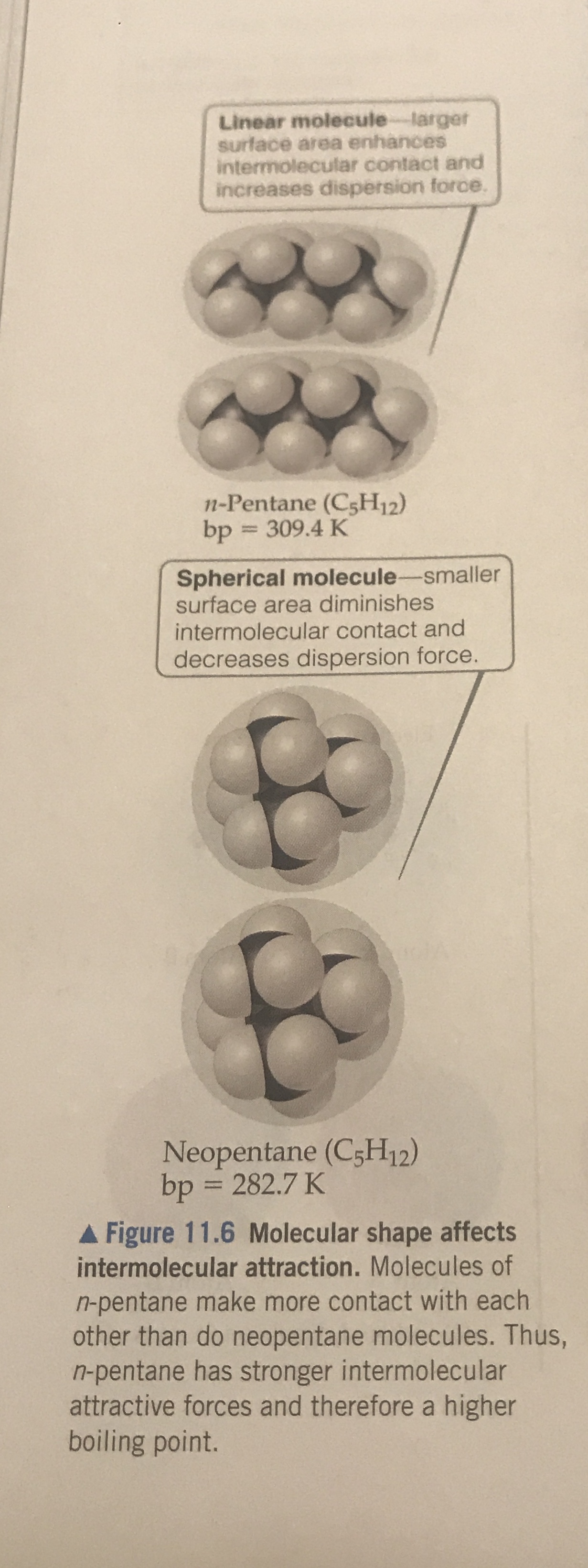
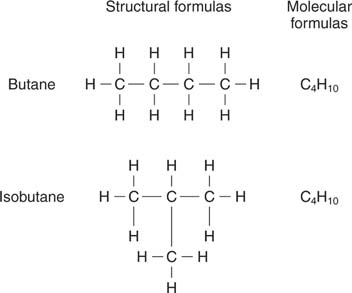
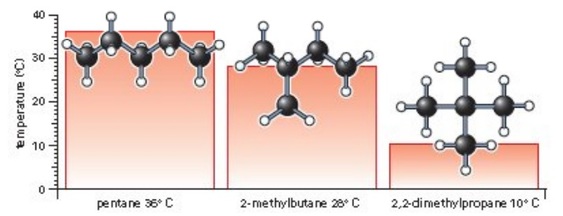
The Effect of Molecular Surface on the Boiling Point-Boiling point increases with surface | Teaching chemistry, Organic chemistry, Chemistry education

Compound C5H12 forms four structurally different monochloroderivatives. C5H12 is therefore: a) n-pentane b) 2-methylbutane c) 2,2-dimethylpropane d) 2,3-dimethylpropane | Homework.Study.com

SOLVED: The strongest intermolecular interactions between pentane (C5H12) molecules arise from London dispersion forces b) hydrogen bonding dipole-dipole forces ion-dipole interactions In which molecule is the central atom sp hybridized? SO2 b)

Drawing 2D structures of different isomers of alkane with molecular... | Download Scientific Diagram

Figure 5. Drawing 2D structures of different isomers of alkane with molecular formula C5H12 and predict their Boiling Point. The student can figure out that branching decreases boiling point. It is worth